 SomaLogic CEO steps down; Q4 revenue declines 18%
SomaLogic CEO steps down; Q4 revenue declines 18%
March 31, 2023 -- Aptamer-based proteomics firm SomaLogic said after the close of the market on Tuesday that its fourth-quarter revenue declined 18% year over year, primarily due to the recognition of $1.9 million of licensing revenue in the fourth quarter of 2021. Read More
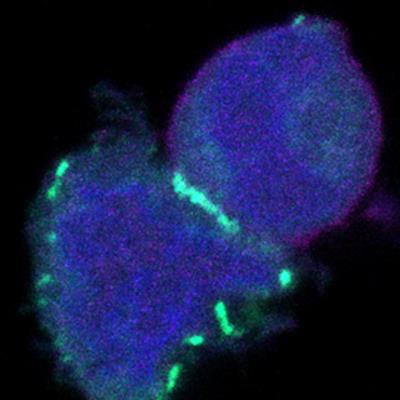 New method detects 'buddy' proteins within cells
New method detects 'buddy' proteins within cells
December 7, 2022 -- Researchers from University of Chicago’s Pritzker School of Molecular Engineering have developed a method to find proteins located in close proximity to each other inside a cell. The technology uses high-throughput genomic sequencing to measure proteins, protein complexes, and mRNA within individual cells. Read More
 Mitochondria seen as common thread in autism spectrum disorder
Mitochondria seen as common thread in autism spectrum disorder
November 28, 2022 -- Researchers in Canada have discovered previously unknown connections between genetic factors in autism spectrum disorder, specifically with the mitochondria. Read More
 Researchers discover way to increase synthetic mRNA's protein production
Researchers discover way to increase synthetic mRNA's protein production
November 23, 2022 -- A team of researchers from the Hong Kong University of Science and Technology has discovered a way to increase synthetic mRNA's protein production efficiency by up to 10 times. They contend that their findings can improve the effectiveness of mRNA vaccines and drugs with even less dosage of the mRNAs. Read More
 Scientists use new chemoproteomic method for global profiling of arginine dimethylation
Scientists use new chemoproteomic method for global profiling of arginine dimethylation
October 20, 2022 -- A study published October 18 in the journal Proceedings of the National Academy of Sciences has revealed how arginine dimethylation regulates protein liquid-liquid phase separation and membraneless organelles by using a new chemoproteomic method. Read More
 Link between circadian disruption and cancer may have to do with temperature: study
Link between circadian disruption and cancer may have to do with temperature: study
September 28, 2022 -- Scientists from the Scripps Research Institute have discovered a surprising link between circadian disruption and cancer that may have to do with temperature. Read More
 Mice study points to glia cells as potential therapy target for Huntington’s disease
Mice study points to glia cells as potential therapy target for Huntington’s disease
August 31, 2022 -- A mice study by University of Rochester Medical Center researchers has reinforced the potential of targeting glia cells -- important support cells found in the brain -- for the treatment of Huntington’s disease, and possibly outright replacement. Read More
 University of Chicago, Kansas awarded $1.2M to transform understanding of RNA splicing
University of Chicago, Kansas awarded $1.2M to transform understanding of RNA splicing
August 29, 2022 -- The W.M. Keck Foundation has awarded a $1.2 million grant to researchers at the University of Chicago and the University of Kansas to help identify human gene sequences that will respond to drug therapies. Read More
 Researchers identify biomarkers resistant to chemotherapy in triple-negative breast cancer
Researchers identify biomarkers resistant to chemotherapy in triple-negative breast cancer
August 25, 2022 -- Researchers have discovered the biological markers in triple-negative breast cancer that are associated with resistance to chemotherapy. Read More
 Seer study shows power of engineered nano-bio interactions, deep access to proteome
Seer study shows power of engineered nano-bio interactions, deep access to proteome
August 24, 2022 -- Life sciences company Seer is highlighting the results of a study detailing how the combination of proteomic methods, nanoengineering, and machine learning enables the capture of thousands of proteins. Read More
Conferences
Member Rewards
Earn points for contributing to market research. Redeem your points for merchandise, travel, or even to help your favorite charity.
Research Topics
Interact with an engaged, global community of your peers who come together to discuss their work and opportunities.
Connect
Tweets by @ScienceBoard



