June 17, 2020 -- Nanoparticles cloaked with human cell membranes can attract and neutralize SARS-CoV-2, according to a new publication in Nano Letters on June 17. The process of binding to the virus hijacks its ability to replicate, making the nanoparticles a viable medical countermeasure against the virus.
The search for new drugs that block viral replication of SARS-CoV-2 requires a clear understanding of underlying molecular mechanisms. Most antiviral drugs target only specific viral species and therefore cannot be deployed across different species or families of viruses and may not be effective as viruses accumulate mutations.
An effective treatment that inhibits SARS-CoV-2 infectivity -- as well as its potential mutated species -- would be valuable in the fight against COVID-19.
Developing cloaked nanoparticles
The new paper describes a novel drug development approach that focuses on affected host cells instead of targeting the causative agent. Here, researchers from the University of California (UC), San Diego created cellular "nanosponges" made of human cell-derived membranes that are naturally targeted by SARS-CoV-2. The nanosponges display the same receptors that the virus depends on for cellular entry.
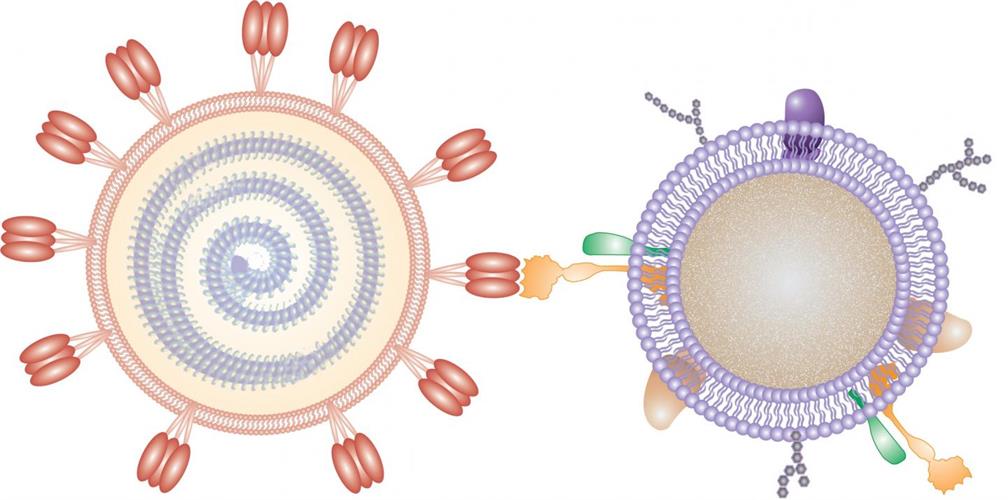
The UC San Diego team first created the membrane-cloaked nanoparticles over a decade ago. Originally, they were developed to treat pneumonia by a San Diego startup, Cellics Therapeutics, which is currently in the process of submitting an investigational new drug application to the U.S. Food and Drug Administration for its lead candidate, with clinical trials expected to begin next year.
For entry into human cells, SARS-CoV-2 uses angiotensin-converting enzyme 2 (ACE2) and CD147 expressed on host cells as receptors. Human macrophages express CD147 and play a role in viral infection by interacting with virus-targeting cells through chemokines and phagocytosis signaling pathways.
Based on this knowledge, the researchers developed two types of cellular nanosponges from human lung epithelial type II cells and human macrophages. The nanosponges were characterized for physiochemical and biological properties and evaluation in vivo and in vitro. The samples were tested in a biosafety level 4 laboratory at Boston University's National Emerging Infectious Diseases Laboratories for inhibitory effects on human SARS-CoV-2 virus and antiviral efficacy.
The nanosponges were constructed by first deriving cell membranes by differential centrifugation that had been verified for purity. The membranes were coated onto polymeric nanoparticle cores made from polylactic-co-glycolic acid (PLGA) with a sonication method. The changes to the core are consistent with the addition of a bilayer cell membrane and the preparation steps enabled membrane protein retention and enrichment on nanosponges.
Western blot analysis revealed the presence of ACE2, transmembrane protease serine 2 (TMPRSS2), and dipeptidyl peptidase IV (DPP4) viral receptors on the epithelial-based nanosponge, and ACE2, C-type lectin domain family 10 (CLEC10), and CD147 on the macrophage-based nanosponge.
To evaluate toxicity after in vivo administration, the researchers dosed mice via the intratracheal route and analyzed histopathological and blood samples. They found that immune infiltration of nanosponges was similar to baseline levels and there was no evidence of lesions or tissue damage. All blood markers were also in line with baseline levels. Cumulatively, this confirms the short-term safety of the nanosponges in vivo.
Lastly, the team evaluated the neutralization of infectivity by SARS-CoV-2 with a plaque reduction neutralization assay. This demonstrated that both types of nanosponges act in a dose-dependent manner to inhibit viral infectivity of SARS-CoV-2 in Vero E6 cells. At a five milliliter concentration, the epithelial-based nanosponge inhibited 93% of viral infectivity and the macrophage-based nanosponge inhibited 88% of viral infectivity.
A potentially virus-agnostic therapy
The researchers emphasize that the nanosponges are mutation- and potentially virus-agnostic. This means that as long as the target of the virus remains the same, the nanosponge will be able to neutralize the infection and potentially provide broad-acting countermeasures against the current virus and future emerging coronaviruses.
"Another interesting aspect of our approach is that even as SARS-CoV-2 mutates, as long as the virus can still invade the cells we are mimicking, our nanosponge approach should still work. I'm not sure this can be said for some of the vaccines and therapeutics that are currently being developed," said Liangfang Zhang, PhD, a nanoengineering professor at the UC San Diego Jacobs School of Engineering.
In the coming months, the researchers will evaluate the efficacy of nanosponges in animal models. The UC San Diego team has already shown short-term safety in the respiratory tracts and lungs of mice. The team also plans to study whether the macrophage sponges also have the ability to quiet cytokine storms in COVID-19 patients.
Do you have a unique perspective on your research related to molecular engineering or virology? Contact the editor today to learn more.
Copyright © 2020 scienceboard.net








