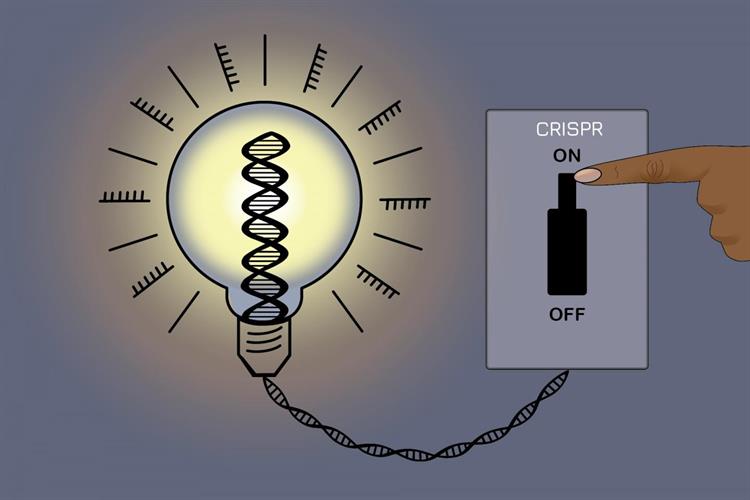
April 12, 2021 -- A new gene silencing tool allows scientists to switch genes on and off without altering genetic sequences. The tool, described in a paper published in Cell on April 9, uses a modified CRISPR-Cas9 system to introduce reversible epigenetic changes that control gene expression.
The classic CRISPR-Cas9 system led to an explosion in gene editing studies since it was first developed almost a decade ago. It became quickly popular thanks to its ease of use compared to previous gene editing methods.
CRISPR-Cas9 uses guide RNA to chaperone the Cas9 nuclease to a particular site in the DNA and create a double-strand break. By adding a specially designed short sequence of DNA, scientists can introduce permanent genetic changes to the cell. This oligonucleotide is used as a template by the cell when repairing the double-strand break, incorporating the sequence into the cell's genome. However, this approach can cause unintended genetic changes.
"As beautiful as CRISPR-Cas9 is, it hands off the repair to natural cellular processes, which are complex and multifaceted," said Jonathan Weissman, PhD, professor of biology at the Massachusetts Institute of Technology, in a statement. "It's very hard to control the outcomes."
To get around this difficulty, Weissman and his colleagues attempted to find a way to control gene expression without altering the genetic sequence. Gene expression is controlled naturally in the cell through epigenetic changes, such as adding methyl groups to the DNA strands. These chemical tags make DNA sequences inaccessible for transcription by RNA polymerase, thus silencing the gene.
Weissman and colleagues saw that they could use this natural process to create a system that would enable them to switch genes on and off.
On and off
The result is a tool called CRISPRoff, which uses an inactive Cas9 protein fused to domains from transcriptional repressor proteins. Guide RNAs can target CRISPRoff to methylate genes of interest and silence them. Another tool called CRISPRon, a fusion protein including transcriptional activator domains, can reverse the process and switch the genes back on.
The team tested the CRISPRoff system in different human cell lines, showing that it could specifically, and almost completely, repress a targeted gene. RNA sequencing experiments on a range of genes affecting growth revealed that the system could be applicable almost across the entire genome.
"The big story here is we now have a simple tool that can silence the vast majority of genes," said Weissman. "We can do this for multiple genes at the same time without any DNA damage, with great deal of homogeneity, and in a way that can be reversed. It's a great tool for controlling gene expression."
A surprising feature is that CRISPRoff was able to silence genes without CpG islands, regions that are rich in repeated cytosine-guanine sequences. CpG islands are found near the transcription start site of most genes and were thought to be necessary for DNA methylation to be effective.
"What was thought before this work was that the 30% of genes that do not have a CpG island were not controlled by DNA methylation," said Luke Gilbert, PhD, an assistant professor at the University of California, San Francisco and co-senior author of the study. "But our work clearly shows that you don't require a CpG island to turn genes off by methylation. That, to me, was a major surprise."
A viable strategy
Previous efforts by the team to create epigenetic editors required cells to continually produce artificial proteins to maintain the changes. The new CRISPRoff system means that the changes are remembered by the cell and passed down through more than 450 cell divisions.
The team tested the technique by silencing a gene in induced pluripotent stem cells. After inducing the cells to become neurons, they found that the gene had remained silenced in 90% of the cells.
"I think our tool really allows us to begin to study the mechanism of heritability, especially epigenetic heritability, which is a huge question in the biomedical sciences," said first author James Nuñez, PhD, a postdoctoral researcher in Weissman's lab.
To demonstrate the potential use of CRISPRoff as a treatment, the team showed that they could use the tool to reduce tau protein being expressed in neurons. Tau, one of the key hallmarks of Alzheimer's disease, accumulates in toxic levels.
"What we showed is that this is a viable strategy for silencing tau and preventing that protein from being expressed," Weissman explained. "The question is, then, how do you deliver this to an adult? And would it really be enough to impact Alzheimer's? Those are big open questions, especially the latter."
The authors also suggested that the CRISPRoff system could help target specific alleles with disease-associated mutations. It could also potentially be used to increase the expression of some genes by silencing the antisense RNA transcripts that inhibit those genes.
"Since we now can sort of silence any part of the genome that we want, it's a great tool for exploring the function of the genome," Weissman concluded.
Do you have a unique perspective on your research related to synthetic biology or gene therapy? Contact the editor today to learn more.
Copyright © 2021 scienceboard.net