March 29, 2021 -- On March 27 the U.S. Food and Drug Administration (FDA) approved idecabtagene vicleucel (Abecma), the first cell-based gene therapy for treating adult patients with multiple myeloma.
Multiple myeloma is an uncommon form of blood cancer in which abnormal plasma cells build up in the bone marrow and form tumors in many bones of the body. The disease prevents bone marrow from making enough healthy blood cells and can damage the kidneys and weaken the immune system. Myeloma accounted for approximately 1.8% of all new cancers in the U.S. in 2020, according to the U.S. National Cancer Institute.
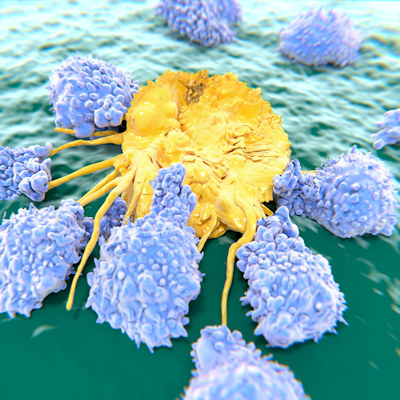
Abecma is a B-cell maturation antigen (BCMA)-directed, genetically modified autologous chimeric antigen receptor (CAR) T-cell therapy. Each dose is customized by using a patient's own T cells, which are collected and genetically modified to include a new gene that facilitates targeting and killing myeloma cells. Once modified, the T cells are infused back into the patient.
The treatment is indicated for patients who have not responded to or whose disease has returned after at least four prior lines of therapy. The safety and efficacy of the product were established in a multicenter study of 127 patients with relapsed myeloma and refractory myeloma, who received at least three prior antimyeloma lines of therapy. In all, 72% of patients partially or completely responded to the treatment and 28% of patients showed a complete response (disappearance of all signs of multiple myeloma).
Due to one of the most serious side effects of the treatment -- cytokine release syndrome -- the FDA is requiring that hospitals and clinics that dispense Abecma be specially certified, and staff are trained to manage cytokine release syndrome, nervous system toxicities, and other side effects. The FDA is also requiring the manufacturer to conduct a postmarketing observational study involving patients treated with the cell-based gene therapy.
The FDA granted approval of Abecma to Celgene, a Bristol Myers Squibb company.
Copyright © 2021 scienceboard.net