November 12, 2020 -- Two groups of researchers have developed unique approaches to overcome the limitations of delivery of gene editing therapeutics. In a pair of new papers, researchers describe methods for more efficient and safe delivery of CRISPR components to targeted cells and tissues.
First, a novel light-activated liposomal delivery system as an alternative to traditional viral vectors was reported in ACS Materials and Interfaces on November 11. The second approach, the use of microRNA (miRNA) to specifically modify expression leading to toxicity of adeno-associated viral (AAV) vectors, was reported in Science Translational Medicine on November 11.
CRISPR has revolutionized the area of precision engineering of genes. The most common version of the system uses a nuclease protein (Cas9) to introduce a double-stranded break in a target sequence of a DNA molecule. This enables the incorporation of a new sequence into the genome as directed by a guide RNA (gRNA) repair template.
The safe and efficient delivery of CRISPR-Cas9 components to targeted cells and tissues remains one of the key challenges for successful gene editing applications. Traditionally, CRISPR relies on viral gene transfer for delivery of components by viral vectors (lentiviral, adenoviral, or AAV) using recombinant DNA technology. However, viral vectors offer only limited spatial and temporal control over the gene editing process, in addition to potential cytotoxicity.
"The traditional delivery vehicle of CRISPR is based on viruses, but they create their own problems because it is difficult to predict the reaction of patients to the viruses," said author Wei Deng, PhD, senior research fellow at the University of South New Wales (USNW) Sydney, in a statement.
Liposomes as a safer alternative
Liposomal formulations for CRISPR-Cas9 systems offer an alternative option for gene delivery. Liposomes -- spherical nanostructures of fat molecules very similar to cell membrane material -- have a high encapsulation capacity and can carry complex cargos. However, the lipid-based nanocarriers are only able to passively release CRISPR-Cas9, suggesting potential issues with spatiotemporal delivery.
Wei Deng's team at UNSW Sydney participated in an international collaborative effort to develop a light-triggered liposome delivery system that can precisely tune the release of gene editing cargo. A clinically photosensitive molecule, verteporfin, was incorporated into the lipid bilayer, and upon light illumination at the 690-nm wavelength, it reacted with available oxygen molecules which rapidly oxidized the unsaturated lipid components. This action destabilized the liposomal structure and released CRISPR.
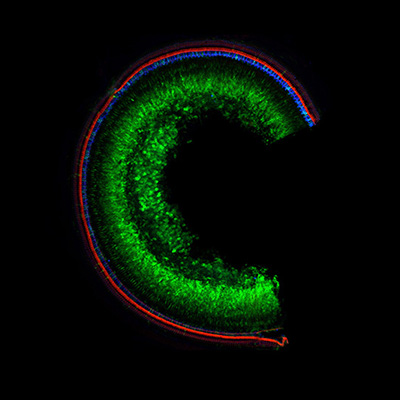
The researchers observed high efficiency of the green fluorescent protein knockout in human HEK293 cells (up to 52%) and in zebrafish embryo models (up to 77%) treated with light-triggered liposomes compared to the control group. Furthermore, the team demonstrated that liposome encapsulation provided sufficient protection for CRISPR to gain cellular entry in cell culture and that the liposome nanoparticles displayed minimal cytotoxicity.
LED light was used to trigger liposomes in both the HEK293 cell lines and zebrafish embryo models. The team found that this activation caused ejection of the CRISPR contents which then go to work looking for the genes of interest. The light can activate the liposomes up to a centimeter below the surface of the skin. While the light-triggered liposomes are limited in tissue penetration, the team plans to study using x-rays to target deep-seated tumors.
"By using x-ray instead of light we also need to find a proper animal model that might translate to using this technology for something like breast cancer treatment," Deng said.
Making viral vectors safer
While the USNW Sydney team is working on alternatives to viral vector delivery of gene editing systems, a group of researchers from Penn Medicine are looking to improve the safety AAV vectors by using miRNA to reduce the toxicity of AAV gene therapy delivered to the central nervous system (CNS).
Most severe toxicities generated from the use of AAV targeting the CNS occur after high-dose intravenous administration. The toxicity has been traced back to the dorsal root ganglion (DRG), which is a cluster of neural cells on the outside of the spinal cord responsible for transmission of sensory messages. The toxicity is associated with degeneration of both peripheral axons of nerves and central axons of the spinal cord.
Recently, the U.S. Food and Drug Administration placed a partial hold on human trials of administering a gene therapy vector into the spinal cord to treat spinal muscular atrophy, the genetic disease that severely weakens muscles and causes problems with movement, due to its toxicity.
The researchers found that the DRG toxicity stems from overexpression-related stress of high intracellular expression of introduced genes, known as a transgene, messenger RNA (mRNA) from AAV vectors. Importantly, the authors noted that adaptive immunity did not play a role in this specific type of toxicity. To correct the toxicity, they developed a strategy to modify transgene expression in DRG neurons using miRNA designed to reduce the level of transgene expression in DRG.
The approach involved cloning miRNA targets that are solely expressed in DRG neurons into the 3' untranslated regions (UTRs) of the transgene. Any mRNA expressed from the vector would be destroyed by the endogenously expressed miRNA.
"We believe it is a safe, straightforward way to ameliorate the safety of AAV therapy for the central nervous system," said first author Juliette Hordeaux, DVM, PhD, senior director of translational research in the University of Pennsylvania Gene Therapy Program, in a statement. "This approach could be used to design other gene therapy vectors to repress transgene expression in the cell types that are affected by the toxicity and not others, which is critical, because you need the expression everywhere else to effectively treat the disorder."
Using the approach, the researchers found that the alteration using an miRNA183 target expression cassette in the vector eliminated more than 80% of the transgene expression and reduced the toxicity in nonhuman primate models. By comparison, unmodified AAV vectors resulted in robust delivery of the gene into target tissues but also toxicity in DRG neurons. Moreover, the addition of miRNA sequences up to 90 days after administration did not affect transduction in other locations in the primate's brain.
"This modified vector shows great promise to reduce DRG toxicity and should facilitate the development of safer AAV-based gene therapies for many CNS diseases," said senior author Dr. James M. Wilson, PhD, director of the gene therapy program and professor of medicine and pediatrics in Penn's Perelman School of Medicine.
"We believe that this new approach could improve safety in gene therapy universally," Hordeaux concluded.
Do you have a unique perspective on your research related to gene therapy or molecular biology? Contact the editor today to learn more.
Copyright © 2020 scienceboard.net