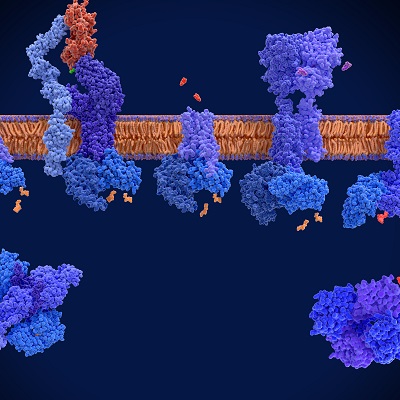
September 1, 2022 -- A new study points to potential new treatments for chronic pain by targeting the metabolite tetrahydrobiopterin (BH4). An international team from the Institute of Molecular Biotechnology of the Austrian Academy of Sciences, Harvard Medical School, and Boston Children’s Hospital, said the therapeutic pathway could improve chronic pain management and combat the opioid epidemic.
Sensory neurons produce the metabolite BH4, which then drives chronic pain such as neuropathic pain or inflammatory pain, and because concentrations of BH4 correlate with pain intensity, the researchers sought to learn more. They performed a phenotypic screen of 1,000 target-annotated, U.S. Food and Drug Administration-approved medications (Science Translational Medicine, August 31, 2022).
The research team started with medications that are currently in use for various indications and tried to identify undescribed, off-target analgesic properties. They linked the previously observed analgesic effects of several drugs, including clonidine and capsaicin, to the BH4 pathway. The phenotypic screen also revealed the drug fluphenazine, an antipsychotic, blocks the BH4 pathway in injured nerves. Researchers also demonstrated its effects in chronic pain following nerve injury in vivo and found the effective dose in mice is comparable to the low end of the dose indicated for schizophrenia in humans.
In addition, the researchers identified a link between the BH4 pathway and EGFR/KRAS signaling, which is involved in multiple cancers. By blocking EGFR/KRAS signaling, pain sensitivity was reduced, as were BH4 levels. Also, by deleting the enzyme GCH1, which is involved in the BH4 pathway, the mouse models of KRAS-driven lung cancer developed fewer tumors and survived much longer.
As a result, the researchers also uncovered a common signaling pathway for chronic pain and lung cancer through EGFR/KRAS and BH4, which could open up new treatment options for both conditions, they contend.
Copyright © 2022 scienceboard.net