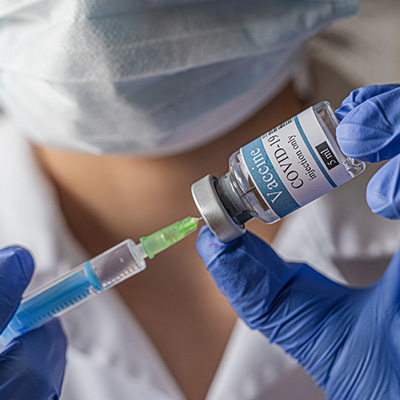
February 16, 2021 -- SK Bioscience is set to supply the Korean government with 40 million doses of Novavax's COVID-19 vaccine through a new collaboration and licensing agreement.
The deal with the Korean government covers the manufacturing and commercialization of NVX-CoV2373, expanding on a prior technology transfer agreement. Doses will be available to citizens in the Republic of Korea this year, the companies said in a statement.
Novovax's NVX-CoV2373 is a protein-based vaccine candidate developed with the company's proprietary recombinant nanoparticle technology. The liquid product is stable with refrigeration, which the company says eases distribution.
Copyright © 2021 scienceboard.net
Member Rewards
Earn points for contributing to market research. Redeem your points for merchandise, travel, or even to help your favorite charity.
Research Topics
Interact with an engaged, global community of your peers who come together to discuss their work and opportunities.
Conferences
Connect
Tweets by @ScienceBoard