August 16, 2021 -- The market for artificial intelligence (AI) in drug development and clinical trials has seen renewed interest in recent months, with total funding in the industry now reaching $10.7 billion, according to a report published by U.K. market insights firm Signify Research.
Following a slower period in 2019 and 2020 on account of market uncertainty and the COVID-19 global pandemic, the industry has seen an uptick in interest and capital funding for both emerging and late-stage vendors (which often cite oversubscribed rounds); $3.6 billion of this sum was raised in 2021 alone.
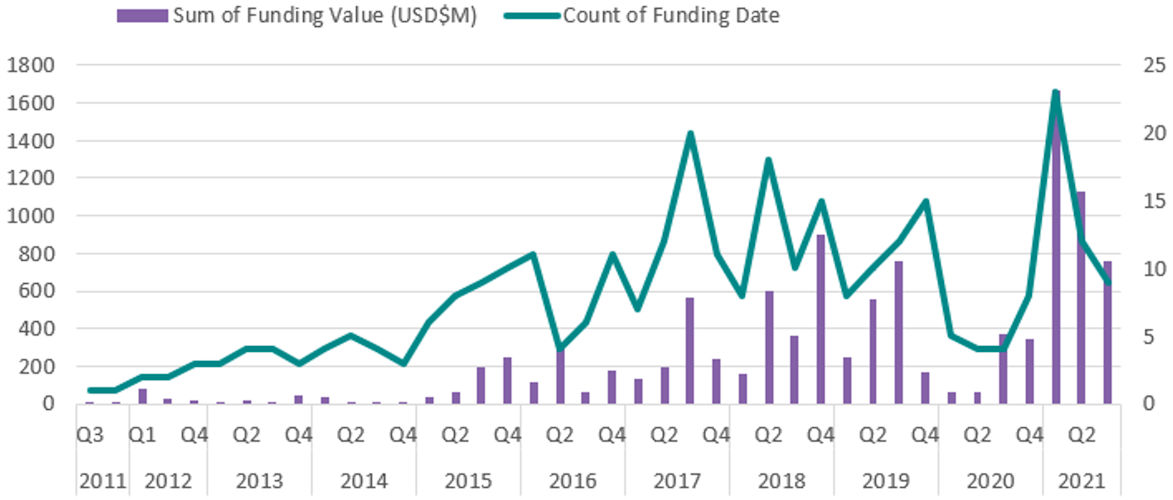
This new funding wave is mostly concentrated around novel drug design companies that offer a high-risk, high-reward potential, signaling a shift in the market as several announced partnerships between pharmaceutical giants begin to reach the five-year mark and see tangible results.
While no novel AI-designed drugs have yet been cleared by the U.S. Food and Drug Administration, the recent pandemic has highlighted the need for a more efficient and dynamic healthcare industry, with many startups benefiting from raising their profiles by participating in COVID-19 research projects. The culture of cooperation has certainly made strides in overcoming barriers that previously held the market back, including promoting and sharing an abundance of data that were previously sequestered by owners.
Freshly launched startups that are emerging from stealth mode will have to enter a market full of well-funded first-generation vendors. The Signify Research team expects to see an increasing number of acquisitions emerge as these well-funded startups look to diversify their portfolios and capitalize on the presence of existing solutions. This will enable acquiring companies to create more beneficial partnerships with existing pharmaceutical companies while improving the quality of internal development pipelines.
As internal pipelines grow, an increasing portion of the market will also pivot toward becoming pharmaceutical "giants" in their own right. We expect an increasing number of initial public offerings to emerge as vendors look to raise the capital required to fund clinical trials.
As these AI companies begin their own clinical trials, several drug design companies will evolve into end-to-end companies also targeting the clinical trials market. By either acquiring or developing their own solutions aimed at reducing the clinical trial timeline, vendors will be able to maximize the efficiency involved in such a lengthy process and hopefully shorten the route to market.
Signify Research is an independent supplier of market intelligence and consultancy to the global healthcare technology industry. Its major coverage areas are healthcare IT, medical imaging, and digital health. Clients include technology vendors, healthcare providers and payers, management consultants, and investors. Signify Research is headquartered in Cranfield, U.K.
The comments and observations expressed do not necessarily reflect the opinions of The Science Advisory Board, nor should they be construed as an endorsement or admonishment of any particular vendor, analyst, industry consultant, or consulting group.
Do you have a unique perspective on your research related to artificial intelligence or drug development? Contact the editor today to learn more.
Copyright © 2021 scienceboard.net






