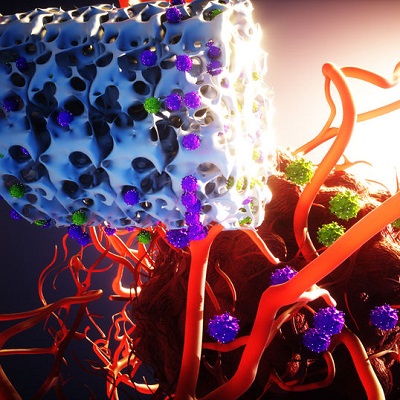
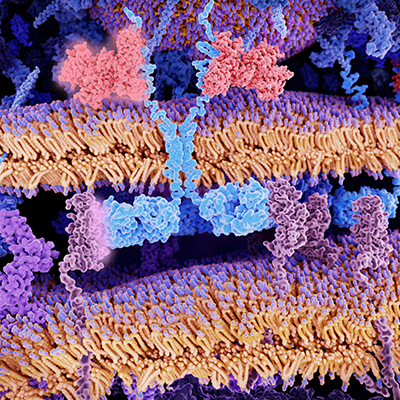
"We need to better understand which patients we should target against. I think the evolution of CAR T is understanding beyond the blood tumors into the solid tumors, and that I think is more challenging," Macrae told ScienceBoard.net at the American Society of Gene & Cell Therapy (ASGCT) 2022 annual meeting in Washington, DC.
However, if the industry is successful with creating allogeneic forms of the therapy "that could be given off-the-shelf" to patients urgently, then cancer could eventually become a "chronic disease of living," Macrae said.
Six CAR T-cell therapies have been approved by the U.S. Food and Drug Administration. All are approved for the treatment of blood cancers. When it comes to the use of CAR T cells to treat solid tumors, advances are hard to come by. However, research on CAR T cells is continuing a pace, including hundreds of ongoing clinical trials.
"Like with all scientific discoveries, there is a moment where everyone thinks it's the best thing since sliced bread -- it'll solve everything. And then, a moment of reality where the despair that it isn't as good as it was thought. And then, a gradual evolution of understanding of which patients it does the best in," Macrae said.
Sangamo has a CAR Treg program, not CAR T. At ASGCT, the company had eight abstracts accepted for presentation, including data from its CAR Treg-cell therapy platform for a first-in-human study involving renal transplantation.Watch the video below to learn more.
Copyright © 2022 scienceboard.net
Last Updated 7/20/2022 2:23:50 PM