 Combination mAbs shown to be effective COVID-19 treatment
Combination mAbs shown to be effective COVID-19 treatment
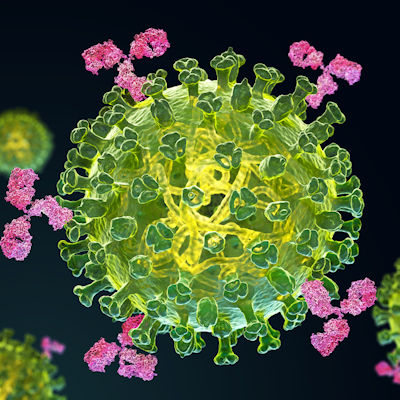
A phase II clinical trial collaboration among Eli Lilly, GlaxoSmithKline, and Vir Biotechnology has shown that a combination of two monoclonal antibodies (mAbs) is effective for treating COVID-19 disease. Read More
 Humanigen's mAb improves outcomes in hospitalized COVID-19 patients
Humanigen's mAb improves outcomes in hospitalized COVID-19 patients
Humanigen is highlighting results from a phase III trial that show that its monoclonal antibody (mAb), lenzilumab, improves outcomes in hospitalized COVID-19 patients by keeping them off mechanical ventilators. Read More
 TCR2 to establish commercial cell therapy manufacturing in Md.
TCR2 to establish commercial cell therapy manufacturing in Md.
TCR2 Therapeutics has signed a long-term lease for an existing 85,000-sq-ft cell therapy manufacturing facility in Rockville, MD, which is ready for current good manufacturing practice build-out. Read More
 Gritstone begins clinical trial of its 2nd-gen COVID-19 vaccine
Gritstone begins clinical trial of its 2nd-gen COVID-19 vaccine
The first patient has been dosed in a phase I clinical study investigating the immunogenicity and safety of Gritstone Oncology's COVID-19 vaccine candidate. Read More
 Cerecor licenses cytokine storm antibody from Kyowa Kirin
Cerecor licenses cytokine storm antibody from Kyowa Kirin
Cerecor has expanded an agreement with Kyowa Kirin for the exclusive worldwide rights to CERC-002, Kyowa Kirin's fully human anti-LIGHT (tumor necrosis factor superfamily member 14, TNFSF14) monoclonal antibody for all indications. Cerecor made the deal through its wholly owned subsidiary, Aevi Genomic Medicine. Read More
 FDA approves 1st cell-based gene therapy for multiple myeloma
FDA approves 1st cell-based gene therapy for multiple myeloma
On March 27 the U.S. Food and Drug Administration (FDA) approved idecabtagene vicleucel (Abecma), the first cell-based gene therapy for treating adult patients with multiple myeloma. Read More
 GSK, Vir file EUA submission for COVID-19 mAb treatment
GSK, Vir file EUA submission for COVID-19 mAb treatment
GlaxoSmithKline (GSK) and Vir Biotechnology filed an emergency use authorization (EUA) application with the U.S. Food and Drug Administration for VIR-7831 (GSK4182136), the firms' investigational dual-action SARS-CoV-2 monoclonal antibody (mAb). Read More
 Bio-Techne launches new product for organoid, stem cell cultures
Bio-Techne launches new product for organoid, stem cell cultures
Bio-Techne has released its new Cultrex UltiMatrix Reduced Growth Factor Basement Membrane Extract (RGF BME) to support organoid and pluripotent stem cell cultures. Read More
 Bruker launches FT-NMR benchtop spectrometer
Bruker launches FT-NMR benchtop spectrometer
Bruker has unveiled its permanent-magnet Fourier 80 system, an 80-MHz Fourier transform nuclear magnetic resonance (FT-NMR) benchtop spectrometer for multinuclear gradient spectroscopy. Read More
 Takeda files dengue vaccine for regulatory approval in EU
Takeda files dengue vaccine for regulatory approval in EU
The European Medicines Agency, an agency in the European Union (EU), has accepted Takeda Pharmaceutical's filing packages for its dengue vaccine candidate (TAK-003), which is under investigation for its ability to prevent dengue due to any dengue virus serotype in individuals ages 4-60. Read More
Member Rewards
Earn points for contributing to market research. Redeem your points for merchandise, travel, or even to help your favorite charity.
Research Topics
Interact with an engaged, global community of your peers who come together to discuss their work and opportunities.
Connect
Tweets by @ScienceBoard



