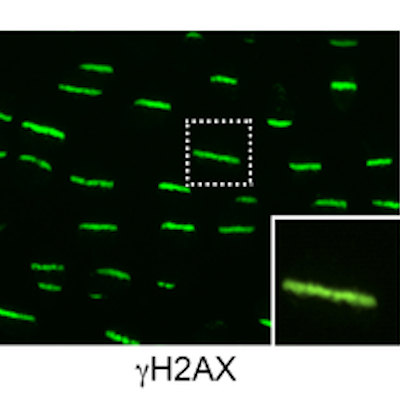
July 20, 2022 -- Researchers have discovered that noncoding "junk" DNA could potentially contribute to the development of cancer. Repetitive DNA sequences can cause replication to stall, triggering a response that is similar to the one induced by DNA damage, according to a study published July 19 in Nature Communications.
The study revealed what happens when replication machinery encounters noncoding junk DNA. While the machinery was able to unwind the DNA strands, it sometimes failed to copy one of the strands, causing replication to collapse.
Because the collapse is similar to what happens after DNA damage, an event known to cause cancer, the researchers believe the noncoding regions of the genome could drive tumor formation.
Studies of neurodegenerative diseases have shown some DNA sequences challenge the replisome, the complex of proteins that enables accurate replication. In conditions such as Fragile X syndrome certain DNA patterns, which are found in unaffected individuals at low levels, are repeated over and over again. Yet, there has been a lack of understanding of how the repetitive sequences impair replication.
Scientists at The Institute of Cancer Research (ICR) in London set out to address that knowledge gap by looking at the process in a test tube, using eukaryotic replisomes assembled from purified proteins to reconstitute DNA replication in vitro. The analysis revealed the point at which the process goes wrong.
When the replication machinery encountered repetitive DNA, it was able to unwind the strands but sometimes failed to copy the opposite DNA strand. The copy failure could cause replication to stall in a manner reminiscent of the response to DNA damage. Kristian Helin, PhD, chief executive of the ICR, set out the implications of the findings in a statement.
"This study helps to unravel the puzzle of junk DNA -- showing how these repetitive sequences can block DNA replication and repair. It's possible that this mechanism could play a role in the development of cancer as a cause of genetic instability -- especially as cancer cells start dividing more quickly and so place the process of DNA replication under more stress," Helin said.
Helin framed the research as part of an ongoing effort to understand the mechanisms underlying genetic mutation and instability. Scientists linked DNA damage to cancer in the 1960s, ultimately leading to the development of poly (ADP-ribose) polymerase (PARP) inhibitors such as AstraZeneca and Merck's Olaparib (Lynparza) that target cancers with DNA repair mutations.
While fresh insights could uncover new ways to exploit the weaknesses of cancer cells, further work is needed to confirm the relevance of the latest findings. The study tested one G4-forming sequence and one i-motif forming sequence that may not represent the behavior of other sequences.
Copyright © 2022 scienceboard.net