December 12, 2019 -- Researchers from the University of Illinois at Urbana-Champaign have successfully used stem cells to engineer nerve tissues as 3D models of neural networks to study brain function. The work was published in the December 3 edition of the Proceedings of the National Academy of Sciences.
Many researchers are working towards the goal of engineering tissues, cellular structures and biological machines in the hopes of recapitulating relevant physiology. These tissue-like structures are an important tool in biomedical applications. The current research is interesting because the scientists have been able to create 3D neural tissues that maintains electrical function.
"If we can control how these neurons communicate with each other, if we can train them using optogenetics, if we can program them, then we can potentially use to perform engineering functions," said Rashid Bashir, professor in the Department of Bioengineering at the Grainger College of Engineering. "In the future, our hope is that by being able to design these neural tissues, we can begin to realize biological processing units and biological computers, similar to the brain."
Current methods of creating 3D neural tissue involve using animal spherical embryoid bodies which have limitations when it comes to scalability on different experimental platforms. The researchers sought to engineer in vitro neural tissues of various shapes and sizes to develop improved models to minimize the use of animal models in biomedical research.
The bio-fabricated neural constructs were developed by leveraging fibrin as a scaffold to engraft differentiated embryonic stem cell-derived neurons. The tissues were formed by differentiation, dissociation and engrafting of neurons in a fibrin hydrogel. The molds were printed by SLA 3D printing techniques, forming the structures for the rod, cube, and toroid shapes. This allowed them to build untethered and geometrically stable in vitro neural tissue mimics that could be molded into various shapes. Moreover, the researchers human stem cells mixed with fibrin can be differentiated in a 3D printed mold, resulting in the formation of a neural tissue mimics.
"It's a bundle of hundreds to thousands of microns of cells that contains a lot of populations with a genetic makeup similar to in vivo tissues," said Gelson Pagan-Diaz, first author of the study. "As we continually develop these bio-fabrication methods, we should be able to capture a lot of the phenomena that happens in vivo. Once we can prove that, we will be able to mimic the morphology that we see in the brain. Once we show that the tissue-engineered outside the body is similar to the tissue in the body, then we can then fabricate them over and over again."
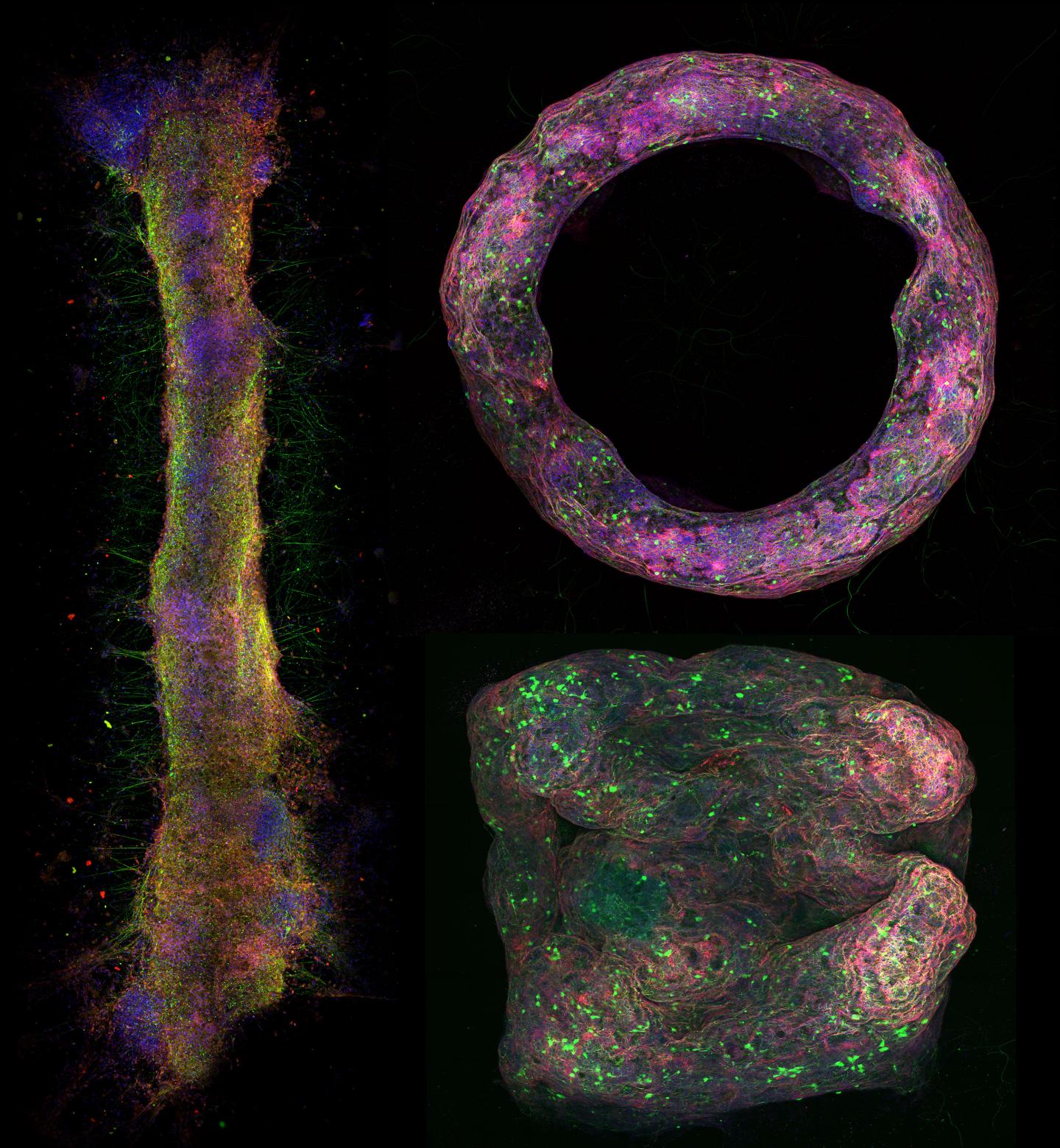
A combination of confocal microscopy and light-sheet microscopy validated that the researchers formed various shapes of neural tissue mimics. Confocal imaging also validated that the neural tissue mimics had normal gene expression and phenotype expression. Optogenetic analysis showed that the mimics were spontaneously active and connected across the variety of shapes. Using the human cell neural mimics, the researchers showed that the constructs responded to drugs to validate its translational application.
"Being able to fabricate these tissue mimics outside the body allows us to characterize and study their electrical activity in great detail," Pagan-Diaz emphasized. "the broad set of design rules due to the 3D structure and shapes gives you a lot more experimental freedom and open up new avenues of research in neuroscience, medicine, and engineering applications."
The researchers believe that these constructs can facilitate assembling biological machines, advance methods for assessment of neural functionality in vitro, and support the development of improved models for disease studies.
Do you have a unique perspective on your research related to 3D printing and neuroscience? Contact the editor today to learn more.
Copyright © 2019 scienceboard.net






