August 5, 2021 -- Biophysics researchers have applied the concept of topological defects to investigate the spread of ovarian cancer cells. Using an in vitro model, the researchers found that these defects -- or disruptions in the normal cellular layout -- can affect the rate of tumor cell invasion. Their findings were published online in APL Bioengineering on August 3.
Topological defects are well known to condensed matter physicists and even cosmologists, who invoke the concept to explain stable configurations of matter formed in the very early universe.
Recently, however, the concept has started to be applied in medicine and biology to explain the density and behavior of configurations of cells in terms of discontinuities in the orientations of cells at specific locations.
In this study, researchers from the University of Wisconsin wanted to explore if (and how) this phenomenon affects metastasis in ovarian cancer. This cancer is of particular interest because it tends to evade detection and present after metastatic spread. Ovarian cancer causes more deaths than any other gynecologic cancer in the U.S., according to the Centers for Disease Control and Prevention.
"My lab is very interested in identifying ways to slow metastasis. This study is exciting because it demonstrates a unique role for organization of nontumor cells to either aid or slow that process," said author Pamela Kreeger, PhD, of the department of biomedical engineering at the University of Wisconsin-Madison, in a statement. "Identifying factors that regulate this organization could help us to achieve our goal."
The researchers studied human mesothelial cells, which is the predominant cell type that covers structures inside the abdomen, where ovarian cancer often metastasizes. Typically, mesothelial cell tissue exhibits a tightly packed "honeycomb" structure.
"A common way to fill space is a honeycomb-like packing in which each 'cell' would be nearly spherical," said author Jacob Notbohm, PhD, an assistant professor in the department of engineering physics at the University of Wisconsin-Madison. "But in our case, the mesothelial cells were elongated, making the honeycomb packing not possible."
The researchers examined a line of mesothelial cells with an elongated, rather than spherical, structure. This elongated structure led to areas of well-ordered cell layers and left other areas with alignment imperfections, causing the topological defects.
Next, the researchers seeded ovarian cancer cells from three different cell lines (OVCAR8, OVCAR3, and OV90) on top of the mesothelial cell layer and examined what effect the arrangement of the mesothelial cells had on how the tumor cells passed through this barrier.
They labeled the respective cells with color and used time-lapse fluorescence microscopy to image the normal cells and cancer cells independently. The researchers tracked the movement of the cells with a transwell assay. The imaging showed the cancer cells penetrating the mesothelial layer over a period of several hours, clearing away the mesothelial cell layer to create free space that was filled by the spreading cancer cells.
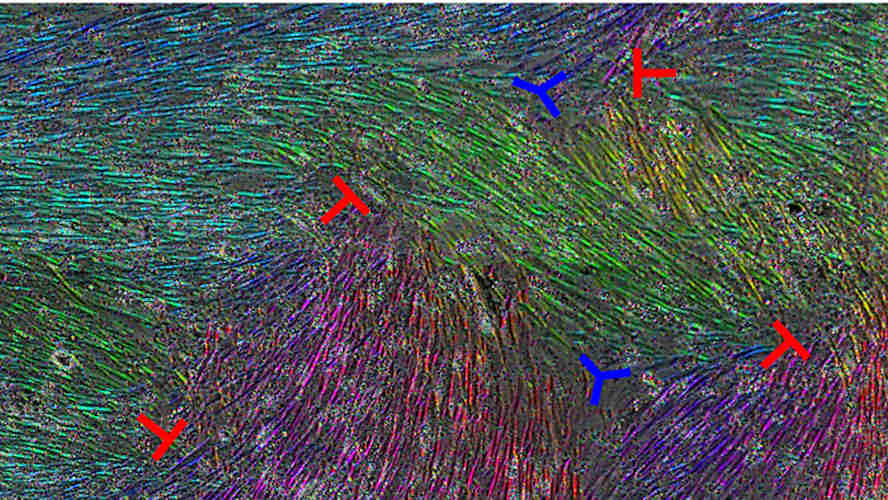
The patterns of cell flow were different near the topological defects, with certain defects causing inward cell flow toward the center of the defect. At those locations, the cancer cells passed through the mesothelial barrier more slowly. In other words, the collective behavior of the mesothelial cells, as governed by the topological defects, was able to slow the spread of the tumor cells. These findings were consistent across all three ovarian cancer cell lines.
In future work, the team hopes to investigate the cause of topological defects and to further study the role of topological structure in cancer cell metastasis.
Do you have a unique perspective on your research related to cancer research? Contact the editor today to learn more.
Copyright © 2021 scienceboard.net