February 23, 2021 -- Automated flow cytometry workflows outperform manual gated techniques by reducing variability among samples, while still achieving a high degree of accuracy. Automated workflows offer a number of advantages to pharmaceutical companies that can integrate them to accelerate drug discovery and development.
As the growing global demand for new drugs soars, the need for efficient analysis of preclinical and clinical research data is a priority for pharmaceutical companies looking to expand their drug development efforts. It is vital that companies generate evidence to show that a new therapy provides an improvement on the current standard of care.

Flow cytometry is one way to address this requirement that can potentially increase pharmaceutical pipeline generation and management. But flow cytometry measures dozens of parameters of cells, which means the data generated are vast and require significant computational power. Therefore, data exploitation is a crucial strategy to utilizing flow cytometry in pharmaceutical drug development.
Ensuring this data are valuable can therefore be a challenge for biopharmaceutical companies and is often time-consuming and inefficient. With this in mind, researchers must turn to tools and technologies that unlock their data and help them to better understand disease behaviors and thus develop new drugs.
Moving away from manual gating
There are several steps during the flow cytometry data life cycle, including processing. This is built on the principle of gating, which groups cell populations based on common characteristics, and is necessary for the visualization of correlations in multiparameter data.
The standard processing method used by many laboratories is manual gating. This means that processing takes around 15-30 minutes and transferring data needs to be managed manually through software packages and people. It is a lengthy process with no guarantee of valuable insights following the hard work undertaken by analysts and scientists.
Alternatively, scientists can instead turn to technology that helps streamline their data extraction and analysis. For example, automated gating offers a new computational approach for flow cytometry processing, with significant gains compared to the manual method.

The advanced technique offers great advantages to scientists when processing their data, including reducing the hands-on processing time for every analysis while retaining control. Through using supervised machine-learning algorithms to apply gates individually for each file, automated methods can mimic human gating strategies. Cumulatively, the automated process is more efficient in reducing time-spent and subsequently reduces overall costs.
Quality control is critical in highly regulated environments when data analysis is at stake. During manual gating, quality control is impractical for large-scale analysis due to the sheer volume of data. Automation of data analysis allows for computational filtering of individual files for low quality data, plus the identification and normalization of batch effects across larger datasets.
As with quality, data reproducibility is a key aspect of pharmaceutical research that can be compromised by manually processing data, as a result of subjectivity. Automated gating has an extremely high reproducibility rate -- unwanted variation is removed -- and consistency is increased during every step, from raw data to results. This means that valuable insights are easier to extract.
Facilitating data sharing
The automated process also facilitates a collaborative approach to data sharing, where data, pipelines, and current work can be shared within and outside of groups and organizations, with varying levels of permissions for data access. This allows the often-difficult process of sharing data through different platforms to be managed in one space. Ultimately the automated workflow boosts both productivity and efficiency, to gain valuable insights at a faster pace than before.
Data can also be uploaded in bulk and gating pipelines can be run in parallel. The supervised gating algorithms are optimized for performance, reducing the file running time to as little as just 30 minutes. This removes the challenges and labor-intensive work of manually gating each file.
Case example: manual versus automated
The advantages of automated gating over manual approaches are highlighted in figure 1. As an example of the power of automated gating, researchers from Aigenpulse compared 210 samples with 39 gates each (8,190 data points), which were gated both manually and automated (using CytoML, Aigenpulse).
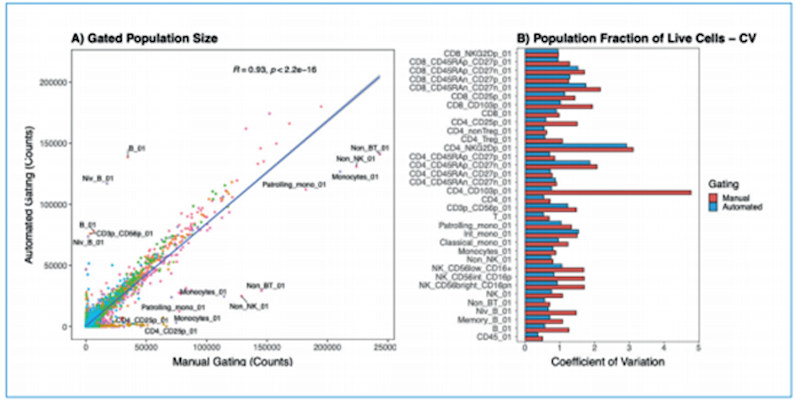
(A) Direct comparison of population sizes from 210 samples gated using manual or automated strategies. Colors represent gated populations. Covariance was measured with Pearson's correlation coefficient. (B) Coefficient of variation for gated populations (normalized to live cells), comparing manual and automated gating strategies. Image courtesy of Aigenpulse.
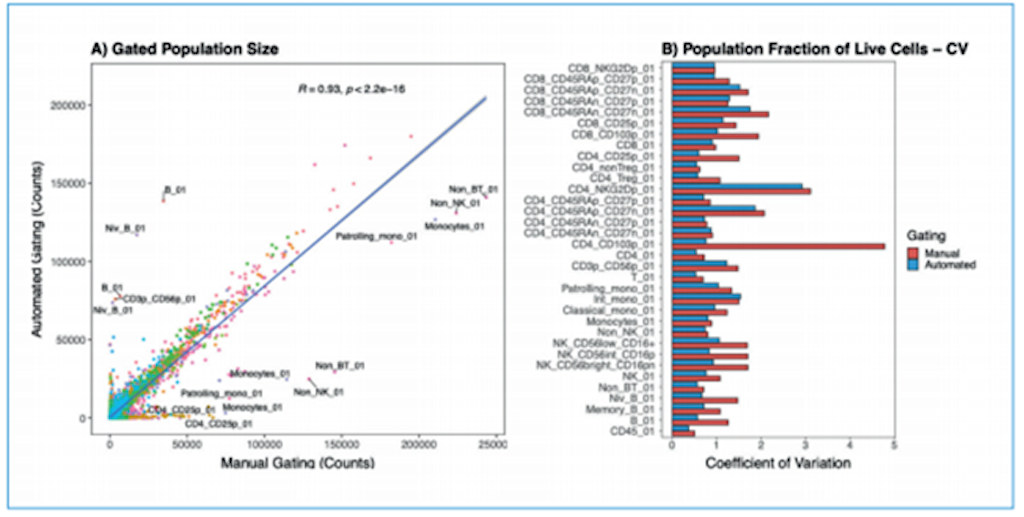
(A) Direct comparison of population sizes from 210 samples gated using manual or automated strategies. Colors represent gated populations. Covariance was measured with Pearson's correlation coefficient. (B) Coefficient of variation for gated populations (normalized to live cells), comparing manual and automated gating strategies. Image courtesy of Aigenpulse.
A strong positive linear correlation (R = 0.93) indicated that the two strategies are highly comparable. Further analysis of the variation between samples for each gate demonstrated that manual gating can be very inconsistent and highly variable, with coefficients of variation greater than 1. This variability was reduced by implementing an automated gating strategy utilizing supervised machine learning.
This demonstrates that a high level of accuracy (equivalent to that of manual gating) can be achieved using automated approaches.
The future is automated
For biopharmaceutical companies that have the capacity to implement changes within their laboratory, switching to an automated process in flow cytometry has vast potential. The technology can help to expedite drug discovery and development, but it is vital that such labs are able to keep up with automation innovation to gain the most out of the data.
Information integration plays a huge role in breaking through the barriers to innovation in the laboratory and allows those labs to scale up their data processing capabilities in an efficient and cost-effective way, saving time and increasing reproducibility of data. Automation provides the industry with tools to find valuable insights from their data that is transparent, accurate, and highly reproducible.
This supports the entire drug discovery process, accelerating and cementing the evidence that drug developers must provide, while also increasing the amount of vital new drugs to those in need.
Satnam Surae, PhD, is the chief product officer at Aigenpulse. Originally focusing in biochemistry, Satnam has now been active in the life sciences for more than 10 years.
Tariq Khoyratty is a computational biologist at Aigenpulse, where he works at the intersection of science and technology, driving the development of the Aigenpulse platform.
Aigenpulse was founded in May 2016 to develop the unique Aigenpulse IT platform that takes into account the dynamic and evolving nature of research.
The comments and observations expressed are those of the authors and do not necessarily reflect the opinions of ScienceBoard.net.
Do you have a unique perspective on your research related to bioinformatics or cell biology? Contact the editor today to learn more.
Copyright © 2021 scienceboard.net







