July 15, 2021 -- Newly designed nanoparticles containing iodine have been shown to kill cancer cells by causing DNA damage when activated by radiation. The in vitro findings were published in Scientific Reports on July 14.
Photon-activation therapy (PAT) is being investigated as a potential cancer treatment because of the ability for x-rays to release electrons with DNA-damaging and cell-killing effects.
"Exposing a metal to light leads to the release of electrons, a phenomenon called the photoelectric effect. An explanation of this phenomenon by Albert Einstein in 1905 heralded the birth of quantum physics," said lead author, Fuyuhiko Tamanoi, PhD, Kyoto University Institute for Integrated Cell-Material Sciences (iCeMS) molecular biologist, in a statement.
However, a known concern about cancer radiation therapy is that it is not effective at the center of tumors, where oxygen levels are low due to a lack of penetrating blood vessels. The x-ray irradiation needs oxygen to generate the DNA-damaging reactive oxygen when the x-rays reach the inside of the cell.
Nanoparticles containing elements such as gadolinium, iodine, gold, and silver (Z elements) have been developed for their ability to be easily taken up by cancer cells and localized in the cell nucleus. Moreover, the enhanced permeability and retention effect (EPR effect) allows nanoparticles to accumulate in tumor cells but not healthy cells.
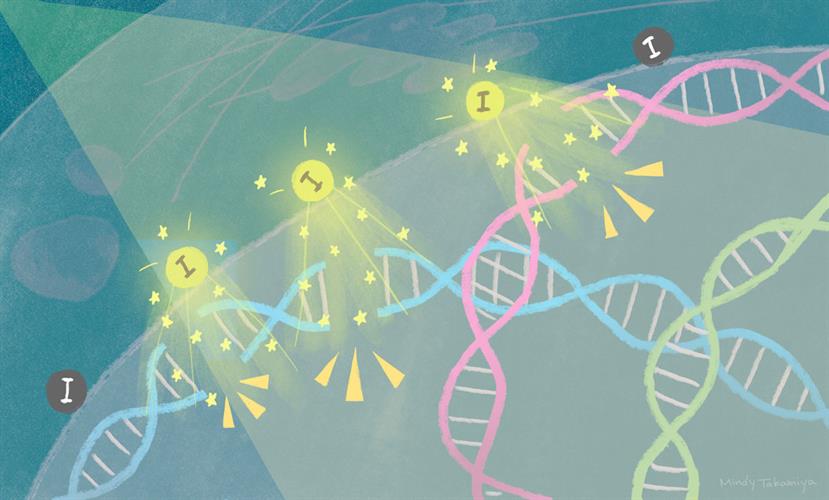
Based on the properties of both radiation therapy and Z elements, and to overcome the limitations of current PAT therapies, a team of Japanese and American researchers designed porous, iodine-carrying organosilica nanoparticles.
The team had previously determined that gadolinium-loaded nanoparticles could kill cancer cells when irradiated with 50.25 keV of synchrotron-generated x-rays. But the iodine used in the current study is cheaper than gadolinium and releases electrons at lower energy levels.
After designing and characterizing the nanoparticles, the researchers dispersed them through tumor spheroids -- 3D tissue containing multiple cancer cells. The spheroids were prepared by growing ovarian cancer cells expressing green fluorescent protein on a special plate that collects cells at the bottom of a well where spheroids are formed.
The team confirmed, by scanning and transmission electron microscopy, that the nanoparticles were efficiently taken up into tumor spheroids and were distributed evenly throughout the spheroids.
Irradiation of the spheroids for 30 minutes with x-rays at an energy level of 33.2 keV led to their complete destruction within three days. This was determined to be the optimum x-ray energy needed to destroy the cancer cells.
Taking the study a step further, the team showed that the iodine-activated nanoparticles caused double-strand DNA breaks in the cancer cells, which subsequently led to apoptosis. Using a fluorescence immunoassay, they determined that the 33.3 keV energy level also provided the most DNA double-strand breaks in the tumor spheroids.
"Our study represents an important example of employing a quantum physics phenomenon inside a cancer cell," said Kotaro Matsumoto, assistant professor at iCeMS. "It appears that a cloud of low-energy electrons is generated close to DNA, causing double-strand breaks that are difficult to repair, eventually leading to programmed cell death."
The team next plans to investigate how electrons are released from iodine atoms when they are exposed to x-rays. They are also working on placing iodine on DNA rather than near it to increase efficacy, and to test the nanoparticles on mouse models of cancer.
Do you have a unique perspective on your research related to cancer therapy or nanoparticle drugs? Contact the editor today to learn more.
Copyright © 2021 scienceboard.net