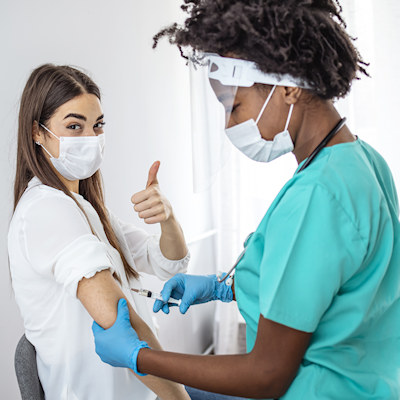
March 12, 2021 -- Johnson & Johnson has been issued an emergency use listing (EUL) by the World Health Organization (WHO) for the Janssen single-shot COVID-19 vaccine to prevent COVID-19 in individuals 18 years of age and older.
The EUL was supported by data from the phase III Ensemble clinical study, showing that the vaccine was well tolerated and provided a 67% decrease in symptomatic COVID-19. The study also demonstrated that the vaccine was 85% effective in preventing severe disease across all regions and protected against different variants.
The EUL procedure streamlines the process by which new or unlicensed products can be assessed for use during public health emergencies by governments and United Nations procurement agencies. The EUL is also required to supply vaccines to the Covax facility for the distribution of COVID-19 vaccines to the participating 190 countries.
In December 2020, the company entered into an agreement with Gavi, the Vaccine Alliance, to support the Covax facility. Johnson & Johnson expects to enter into an advance purchase agreement that would provide up to 500 million doses of the vaccine to Covax through 2022.
Copyright © 2021 scienceboard.net