September 30, 2022 -- Mutations that support the growth of tumor cells may also inhibit T cells, preventing the immune system from responding effectively to emerging cancers, according to Harvard Medical School researchers.
Their findings, published September 29 in the journal Science, provide insights into how certain cancers disarm the immune system so that tumor cells can grow and divide -- with the potential to inform the development of better, more targeted therapies.
Previous studies have found mutations in isocitrate dehydrogenase (IDH), which occur in around 3.5% of cancers, cause the secretion of a metabolite not normally found at high levels in the human body. The metabolite, D-2-hydroxyglutarate (D-2HG), alters the genetic pathways of tumor cells to make them more aggressive and thereby supports the growth of cancers including gliomas and acute myeloid leukemia.
A team led by researchers at Harvard Medical School sought to understand how D-2HG affects other cells in the tumor microenvironment, a region that can be highly immunosuppressive and therefore limit the effectiveness of immuno-oncology therapies such as checkpoint inhibitors and CAR T-cell therapies.
To better understand the role of D-2HG in the tumor microenvironment, the researchers ran a series of experiments in mouse models. The experiments showed that CD8+ T cells sense D-2HG and take it up. When exposed to high levels of the metabolite produced by a tumor, the proliferation of the immune cells slowed down and their cancer-killing power waned.
The effects stem from the inhibition of lactate dehydrogenase, a key metabolic enzyme that plays a role in producing cytokines and granzymes, by D-2HG. The enzyme helps T cells to proliferate and maintain their cancer-killing capacity. While inhibiting the enzyme limited its effects on T cells, and by extension the ability of the immune system to suppress tumors, the T cells regained their ability to kill tumor cells when D-2HG was removed. The result suggests the effects of D-2HG are reversible.
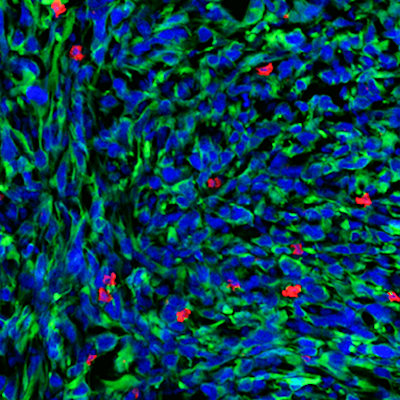
In another set of experiments, the collaborators found that the regions of IDH-mutated human glioma tumors with higher D-2HG levels had lower levels of T-cell infiltration, and vice versa. The findings support the mouse data.
While the study highlights an immune component in this type of cancer that wasn't fully appreciated before, what the findings mean for efforts to tackle immunosuppression in the tumor microenvironment is less clear, explained Marcia Haigis, PhD, senior author and cell biology professor in the Blavatnik Institute at Harvard Medical School, in a statement.
"The field has initially focused on tumor cell functions of this metabolite, and I think that the door is now open for other studies to look at how it impacts immune cells and the whole microenvironment. We still don't know the therapeutic implication of this research -- do IDH inhibitors work in part by increasing the activity of the immune system, or do they only act directly on the cancer cells?" Haigis said.
Copyright © 2022 scienceboard.net