July 1, 2020 -- A surprisingly simple approach to manufacturing silica nanoparticles by taking advantage of fluid dynamics in tiny Teflon tubes could reduce manufacturing costs. The work is described in a June 28 article published in the Chemical Engineering Journal.
In bioprocessing applications, silica gels, beads, or resins are commonly used in separation processes (chromatography) to purify high-value compounds in large quantities. Silica-based nanoparticles are used as excipients in pharmaceutical technology, particularly as drug delivery systems. Their porous structure enables high drug loading, while their poor water solubility protects drugs from enzymatic degradation.
Synthesis of silica particles began in the 1960s via hydrolysis of alkyl silicates and condensation of silanols and silicic acid in alcohol solution. Traditionally, silica particles are synthesized in large batches. With this process, it is difficult to create specific functionality by controlled doping. Alternatively, microstructured chemical reactors (small-scale microfluidic reactors) can provide better control over reaction rate, although it is often costly and time-consuming to achieve.
Lithographically fabricated microfluidic spiral channel devices have been used to synthesize submicron silica spheres in the past. The devices require extensive fabrication steps requiring photolithography for assembly and are often based on polydimethylsiloxane and glass-based chips which are difficult to make uniformly. On the other hand, helical tubular microreactors offer improved heat and mass transport and enhanced mixing.
Researchers at the University of Melbourne-based ARC Centre of Excellence in Exciton Science, working with colleagues at South China Normal University, explored the use of a simple and inexpensive method to produce uniform silica particles with a controlled size between 100-nm and 600-nm diameter and high yields.
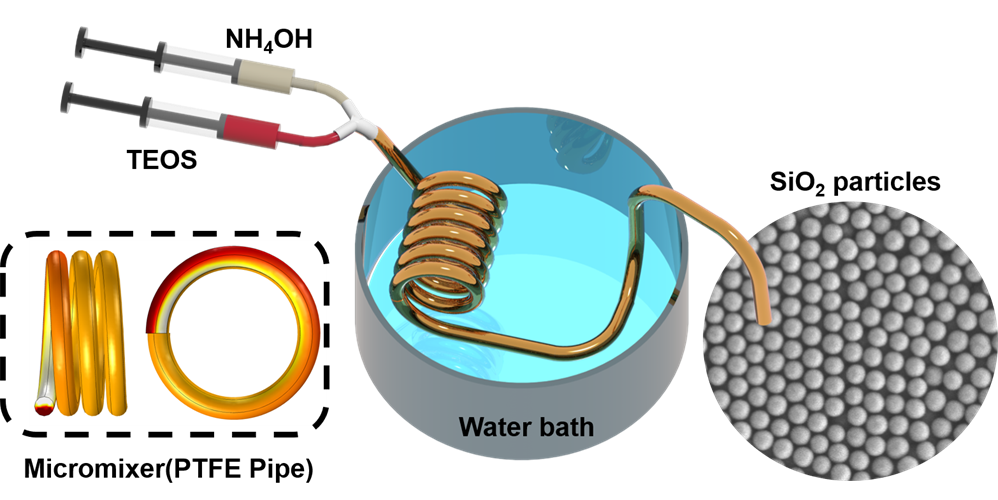
The team constructed a flow-synthesis device using a polytetrafluoroethylene (PTFE) or "Teflon" pipe wound around a rod and connected to two syringes. The new system exploits Dean's flow -- principles that describe the secondary motion of fluid through curved pipes -- to achieve high-throughput and sorting efficiency. In the device, two fluid phases -- ammonia in water and the silica precursor tetraethyl orthosilicate (TEOS) in a nonaqueous solvent -- undergo ultrafast mixing due to Dean's flow effects.
"When this happens you now have this very cheap and quite efficient chemical reactor," said corresponding author Eser Akinoglu, PhD, research fellow at the University of Melbourne, in a statement. "Once you have the fluids in a tube that is spiraled, then due to these vortexes, they mix very quickly and ... the overall reaction is more homogeneous, the product is homogeneous, and the silica particles have a uniform size and shape."
The researchers investigated the effect of different device geometries and process parameters on the production of silica nanoparticle synthesis. They found that the final silica particles produced in various conditions consistently exhibited large negative zeta potentials (electrokinetic potential), indicating strong nanoparticle stability.
By switching from batch-wise to continuous production, the researchers were able to produce uniform particle size with the use of a simple, high-efficiency, high-yielding, low-cost system. While the mechanism may not replace large industrial-scale batch manufacturing, it could be a game-changing approach to the synthesis of silica nanoparticles for specialized, niche applications.
In the future, the researchers plan to demonstrate that the system can be extended to allow doped silica particles to be created.
Do you have a unique perspective on your research related to drug delivery systems or downstream bioprocessing? Contact the editor today to learn more.
Copyright © 2020 scienceboard.net


