December 2, 2019 -- Researchers from Duke Cancer Institute have developed a new technique to observe stem cell mutations occurring in colon cancer.
Details of the technique were published in Nature Communications on December 2.
Somatic mutations in intestinal stem cells within a crypt can lead to colon cancer, occurring slowly over decades. Extreme changes in the intestinal microenvironment can speed the rate of field cancerization (the process by which cells are affected by a carcinogenic alteration). The researchers identified Rspondin-3 (RSPO3) as an oncogenic factor in promoting the spread of cancer within the intestines. However, no models currently exist to investigate the cellular and molecular roles of RSPO3 in field cancerization.
Given the current research limitations, the team at Duke Cancer Institute decided to develop a cancer rainbow (Crainbow) mouse modeling platform which allows them to induce multiple somatic mutations via a cell-labeling technique and visualize changes in intestinal stem cell competition and clone spreading of mutations.
"This study provides new insight into the previously invisible process in which mutant precancerous stem cells spread throughout the colon and seed cancer," said Joshua Snyder, PhD, assistant professor in the departments of Surgery and Cell Biology at Duke University.
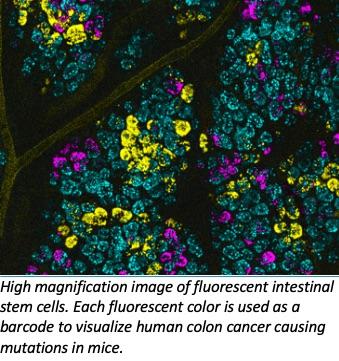
Crainbow is a genetic model system for labeling and visualizing individual cells that express somatic mutations. In the technique there are four positions on the gene that contain either an inert fluorescent protein or three spectrally resolvable fluorescent proteins paired with an oncogenic mutation of choice. This creates a fluorescent barcoding system for tumor driver genes and allows for single-cell resolution of cell fitness, cell signaling and clonal spread of oncogenic mutations. Confocal imaging was used to assess clonal spread.
Lineage dynamics and signaling were assessed by single-cell RNA sequencing (scRNAseq) of crypt enriched tissue isolated from wildtype and precancerous small intestine. The data suggest that microenvironmental oncogenesis extrinsically modifies the stem cell compartment. They also correctly hypothesized that microenvironmental oncogenesis can promote the expansion of oncogenic clones in adult intestines. The findings suggest that stem cell compartments were expanded (RNA FISH and confocal microscopy) and crypt fission was increased (vibratome cross sections and confocal microscopy). Certain common mutations that arise from external sources, such as an injury or an environmental exposure, could also disrupt the environment surrounding the stem cell and lead to the rapid growth and spread of precancerous fields. These events can often be lethal.
"Field cancerization has been suggested to be the defining event that initiates the process of cancer growth, including cancers of the breast, skin and lung," Snyder said. "Our technique allows us to model how premalignant cells compete and expand within a field by simple fluorescent imaging, potentially leading to earlier diagnosis and treatment."
The team is currently using the fluorescent barcoding system to look at other cancers such as breast cancer in the hopes of learning more about pre-cancerous growth and identifying new therapies.
Do you have a unique perspective on your research related to proteomics or cancer? Contact the editor today to learn more.
Copyright © 2019 scienceboard.net


