February 19, 2020 -- New green technology enables accelerated screening and retrieval of "switchable" solvents for chemical and pharmaceutical applications. Utilizing microfluidic strategies, researchers developed an in situ approach to study gas-liquid processes in a time- and material-efficient manner. The work was published online in ACS Sustainable Chemistry and Engineering on February 6.
Solvent extraction is an important part of bioprocess engineering with pharmaceutical applications. It is typical in these industries to use volatile solvents that are easily removed via distillation. However, these solvents are often flammable or toxic, and the distillation process is both energy- and cost-intensive.
Switchable solvents in their original form are hydrophobic (immiscible with water), but they can "switch" to a hydrophilic state (miscible with water) in the presence of water and carbon dioxide (CO2). This unique feature makes them attractive because they can be easily extracted and reused.
However, from an industrial point of view, there are significant challenges, according to Milad Abolhasani, PhD, an assistant professor of chemical and biomolecular engineering at North Carolina State University.
"Specifically, the process for screening candidates to identify the most efficient switchable solvent for a particular application can be extremely time- and labor-intensive," Abolhasani said in a university statement. "And once you have the right switchable solvent candidate, removing it on a large scale can also take a long time."
Traditionally, studies with switchable solvents have been conducted using batch reactors with low extraction rates and long extraction times.
To address these concerns, the research team at North Carolina State University developed a microscale flow chemistry platform that operates on just five microliters of sample. The system contains a tubular CO2-permeable membrane reactor that increases the interfacial area (contact area between two liquids) and continuously delivers CO2 to a reactive switchable solvent mix.
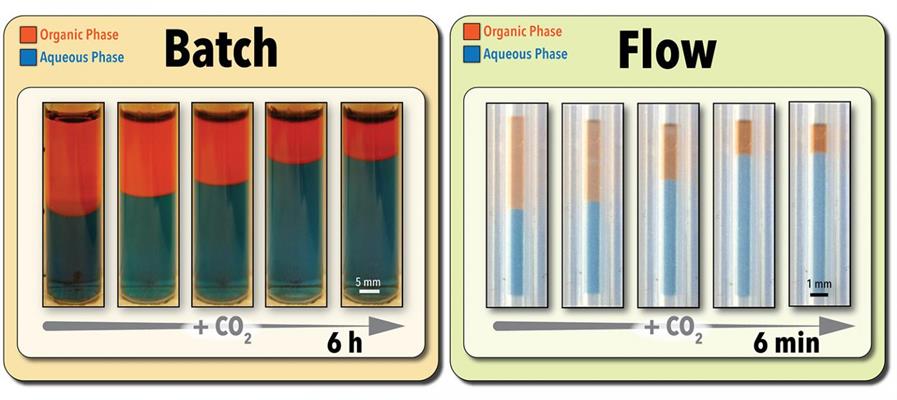
Using the technique, the researchers were able to conduct fundamental studies of continuous factors (reaction time, solvent concentration, CO2 pressure) and discrete parameters (solvent structure).
Using the optimized process parameters as determined by the platform, extraction was achieved in as little as three minutes. Moreover, the system allowed the researchers to run multiple samples simultaneously. This allows users to run approximately 280 screening experiments per day. By comparison, conventional batch extraction processes can take anywhere from six to eight hours -- or approximately one experiment per day.
The team also demonstrated that the flow chemistry technique is just as accurate as conventional batch testing for determining a solvent's efficiency. Therefore, the calculated equilibrium extraction efficiency of switchable solvent in the microscale flow chemistry platform can be directly transferred to large-scale extraction processes. The researchers have recently shown that they can reconfigure the same flow chemistry platform utilized for rapid switchable solvent screening into a continuous flow mode for retrieving solvents on a large scale.
"We're excited about the potential of this process intensification technology and are looking for partners to help us transfer the technique from the lab to industrial R&D and manufacturing applications," Abolhasani said.
Do you have a unique perspective on your research related to downstream manufacturing or chemical engineering? Contact the editor today to learn more.
Copyright © 2020 scienceboard.net


